Diagnostic pathway for suspected AADC deficiency
For patients presenting with some of the key physical symptoms of aromatic L-amino acid decarboxylase (AADC) deficiency, it is necessary to perform a series of diagnostic tests to achieve a definitive diagnosis.1,2
Diagnostic pathway for suspected AADC deficiency2,3
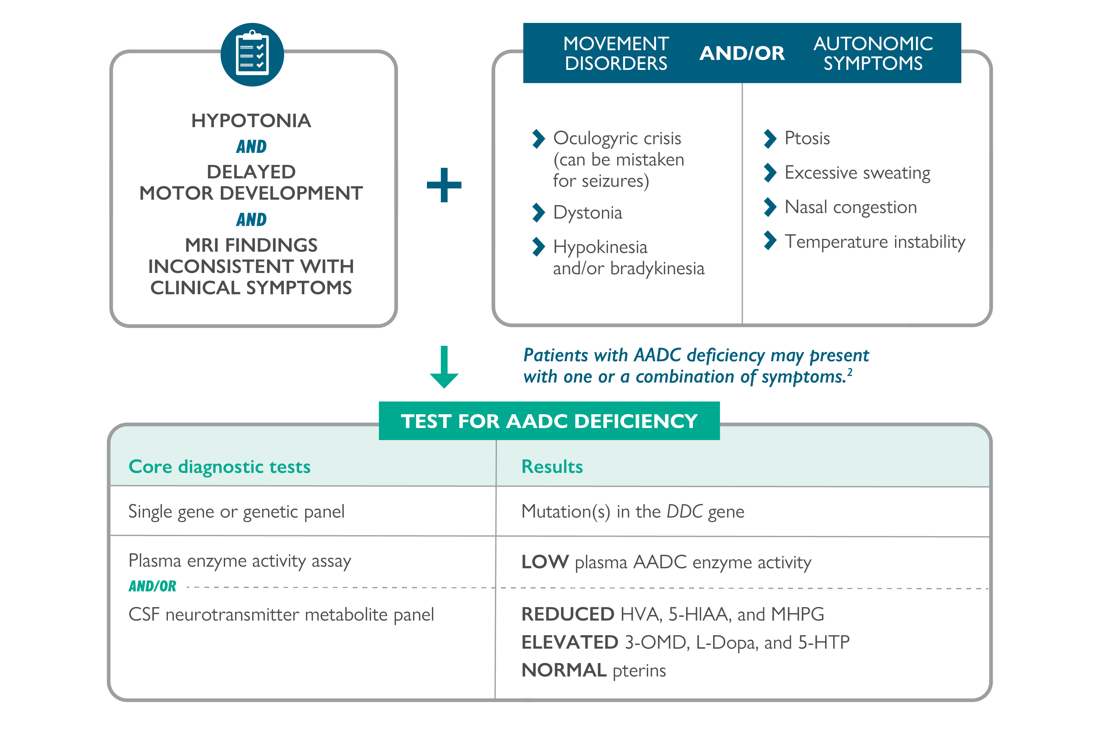
Adapted from Himmelreich, et al 2019.3
3-OMD, 3-O-methyldopa; 5-HIAA, 5-hydroxyindoleacetic acid; 5-HTP, 5-hydroxytryptophan; AADC, aromatic L-amino acid decarboxylase; CSF, cerebrospinal fluid; HVA, homovanillic acid; L-Dopa, L-3,4-dihydroxyphenylalanine; MHPG, 3-methoxy-4-hydroxyphenylglycol.
Screening for AADC deficiency
Newborn screening can offer the opportunity to identify newborns with some inherited or congenital metabolic or endocrine disorders.4 There are currently two screening techniques that can be used to identify suspected cases of AADC deficiency:
- Dried blood spot (DBS) or plasma analysis: Elevated 3-O-methyldopa (3-OMD) concentrations in newborn infants is highly predictive for AADC deficiency and may allow early referral of patients with suspected AADC deficiency for confirmatory testing1
- Urinary testing: Semi-quantitative analysis of urine vanillactic acid (VLA)/vanillylmandelic acid (VMA) ratio is a reliable approach for the screening for AADC deficiency5
Core diagnostic tests
There are three core diagnostic tests for identifying AADC deficiency and confirming a diagnosis:2
- Cerebrospinal fluid (CSF) testing: To measure CSF neurotransmitter metabolites
- Gene sequencing: To check the dopa decarboxylase (DDC) gene for any variants
- Enzyme assays: To measure AADC activity in plasma
CSF testing
CSF evaluation should always include the standard measurements for cells, protein, glucose and lactate, but the following metabolites are markers for AADC deficiency and should be specifically measured:2
- Homovanillic acid (HVA)
- 5-hydroxyindoleacetic acid (5-HIAA)
- 3-O-methyldopa (3-OMD)
- Levodopa (L-Dopa)
- 5-hydroxytryptophan (5-HTP)
- 3-methoxy-4-hydroxyphenylglycol (MHPG)
- Pterins
- 5-methyltetrahydrofolate (5-MTHF), if available
- Pyridoxal phosphate (PLP), if available
The typical CSF profile seen in patients with AADC deficiency shows:2
-
- Low levels of 5-HIAA, HVA and MHPG
- Normal levels of pterins, including neopterin and biopterin
- High levels of 3-OMD, L-Dopa and 5-HTP
Summary of CSF metabolite marker levels typical of AADC deficiency at different ages8
| Marker | Neonatal (birth–1 month) | Infancy (1–18 months) | Childhood (1.5–11 years) | Adolescence (11–16 years) | Adulthood (>16 years) |
|---|---|---|---|---|---|
| 3-OMD | |||||
| L-Dopa | |||||
| HVA | |||||
| 5HIAA | |||||
| 5-HTP | |||||
| MHPG |
*Normal to increased. Adapted from IEMbase Inborn Errors of Metabolism Knowledgebase.8
To ensure correct interpretation of results, the collection and handling of CSF should be performed strictly following standardised procedures.2
Gene sequencing
For the majority of patients, AADC deficiency can be confirmed using genetic sequence and variant analysis.2 Over 100 disease-causing variants in the DDC gene leading to AADC deficiency have been identified to date.3,9–12 The most frequent variants reported include:
- c.714+4A>T3
- p.S250F3
- p.R347Q3
- p.G102S3
- c.1234C>T9,10
- IVS6+4A>T10,11
An up-to-date list of DDC variants can be found on the Pediatric Neurotransmitter Disease database (PNDdb), available at http://www.biopku.org/home/pnddb.asp.
Enzyme assays to measure AADC activity
Severely decreased AADC activity in plasma indicates a diagnosis of AADC deficiency.2 Enzyme assays measuring AADC activity in plasma can be performed using both L-Dopa and 5-HTP as a substrate, but L-Dopa gives a higher analytical yield, so is the standard method.2
Summary
To confirm a diagnosis of AADC deficiency, at least two out of three of the following diagnostic tests should be positive, and genetic testing should be performed:2
- Low CSF levels of 5-HIAA, HVA, and MHPG, increased CSF levels of 3-OMD, L-Dopa, and 5-HTP, and normal levels of CSF pterins
- The presence of compound heterozygous or homozygous pathogenic variants in the DDC gene
- Decreased AADC enzyme activity in the plasma
- If genetic testing is performed as the first step, functional confirmation should be completed by AADC enzyme plasma testing and/or CSF neurotransmitter metabolites panel2
- Chien YH, et al. Mol Genet Metab. 2016;118:259‒263.
- Wassenberg T, et al. Orphanet J Rare Dis. 2017;12:12.
- Himmelreich N, et al. Mol Genet Metab. 2019;127:12‒22.
- Shulze A, et al. Newborn Screening. In: Sarafoglou K, Hoffmann G, Roth K eds. Pediatric Endocrinology and Inborn Errors of Metabolism. 2nd ed. New York: McGraw-Hill; 2017.
- Brennenstuhl H, et al. Mol Genet Metab. 2020;131:163–170.
- Brennenstuhl H, et al. J Inherit Metab Dis. 2020;43:602–610.
- Lee HC, et al. Clin Chim Acta. 2012;413:126–130.
- IEMbase© Inborn Errors of Metabolism Knowledge base. Aromatic L-amino acid decarboxylase deficiency. Available at: http://iembase.org/app/#!/disorder/100. Accessed April 2022.
- Pearson TS, et al. J Inherit Metab Dis. 2020;43:1121–1130.
- Dai W, et al; on behalf of the Mainland Chinese League of AADC Rare Disease. Mol Genet Genomic Med. 2020;8:e1143.
- Monteleone B, Hyland K. BMC Neurol. 2020;20.
- Micallef J, et al. Neuropediatrics. 2020;51:229‒232.
Explore interactive case studies
GL-AADC-1024 | April 2022
